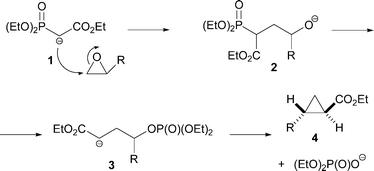
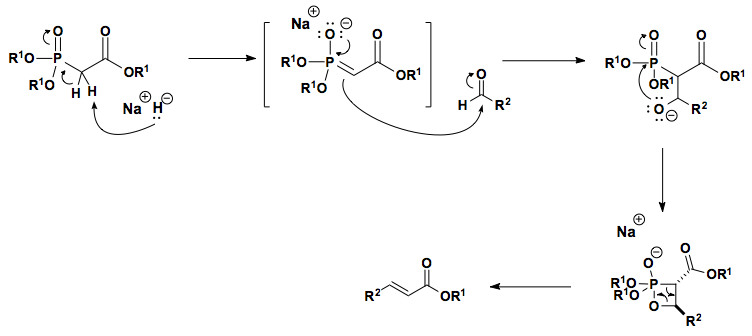
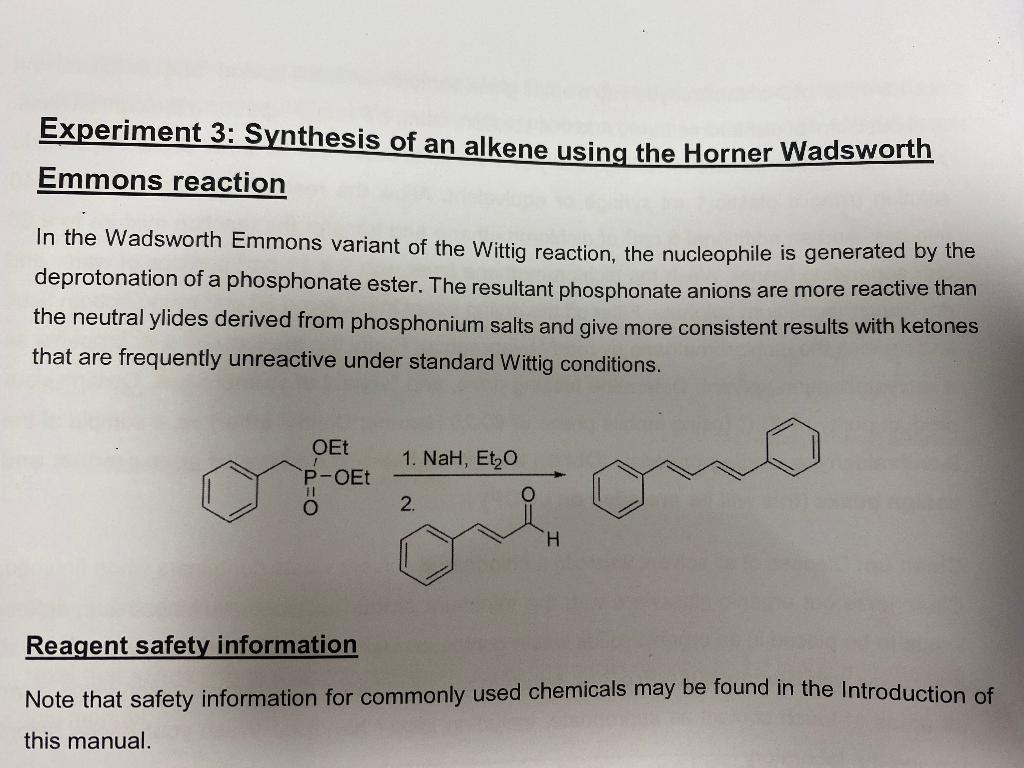
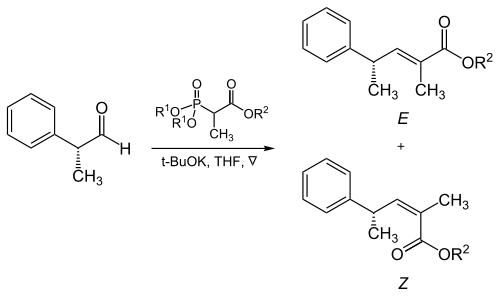
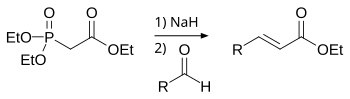
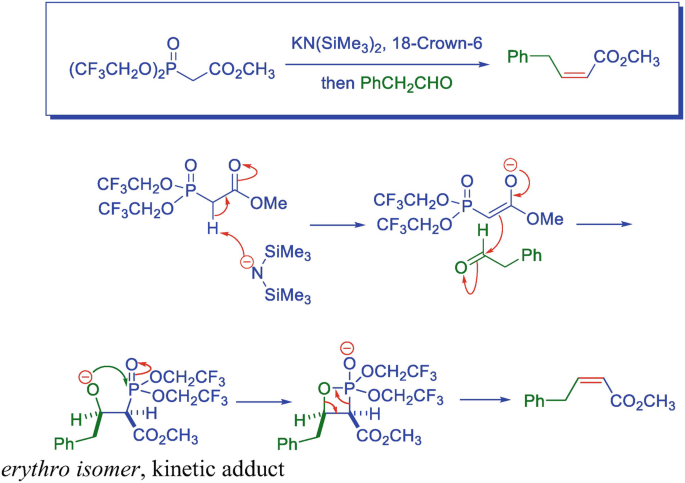
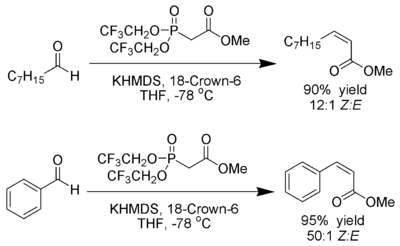
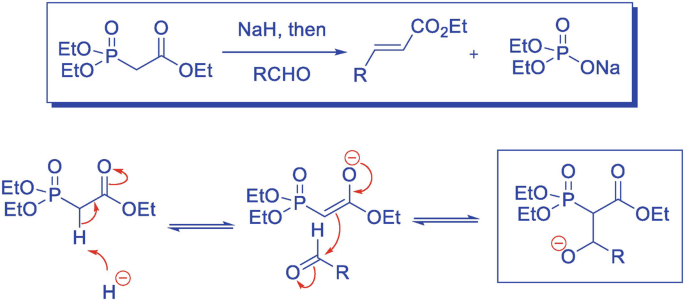
![Roman A. V. on Twitter: "[76] Horner-Wadsworth-Emmons (HWE) Olefination 1958 - compare to Wittig Reaction ⚗️: #100MustKnowMechanisms ➡️ made ⚙️🛠 with @ChemDraw https://t.co/FrXJXfwpOj" / Twitter Roman A. V. on Twitter: "[76] Horner-Wadsworth-Emmons (HWE) Olefination 1958 - compare to Wittig Reaction ⚗️: #100MustKnowMechanisms ➡️ made ⚙️🛠 with @ChemDraw https://t.co/FrXJXfwpOj" / Twitter](https://pbs.twimg.com/media/DXDqPE7W4AE_H2i.jpg:large)
Roman A. V. on Twitter: "[76] Horner-Wadsworth-Emmons (HWE) Olefination 1958 - compare to Wittig Reaction ⚗️: #100MustKnowMechanisms ➡️ made ⚙️🛠 with @ChemDraw https://t.co/FrXJXfwpOj" / Twitter

Synthesis of Butenolides via a Horner–Wadsworth–Emmons Cascading Dimerization Reaction | The Journal of Organic Chemistry
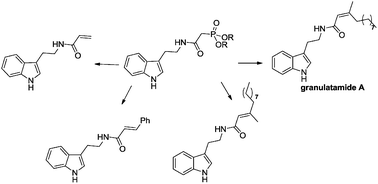
Application of Horner–Wadsworth–Emmons olefination for the synthesis of granulatamide A, its E isomer and other amides of tryptamine - New Journal of Chemistry (RSC Publishing)

Stereoselective Synthesis of Bisfuranoxide (Aurochrome, Auroxanthin) and Monofuranoxide (Equinenone 5′,8′-Epoxide) Carotenoids by Double Horner– Wadsworth–Emmons Reaction | Journal of Natural Products