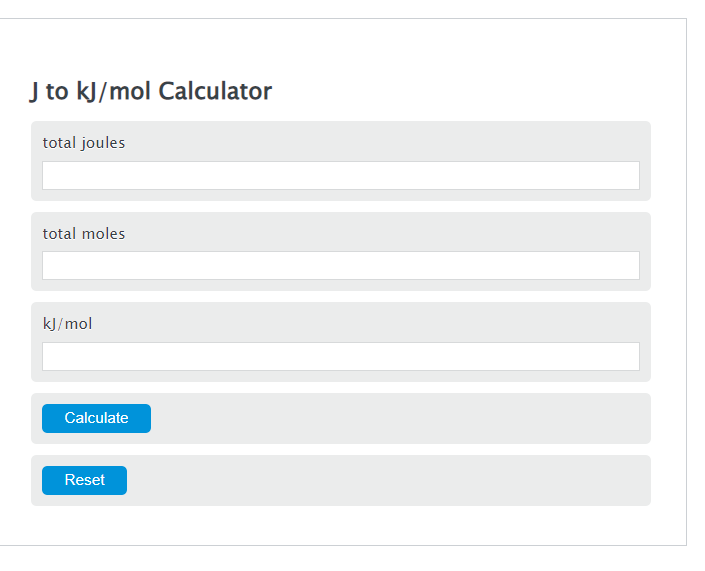
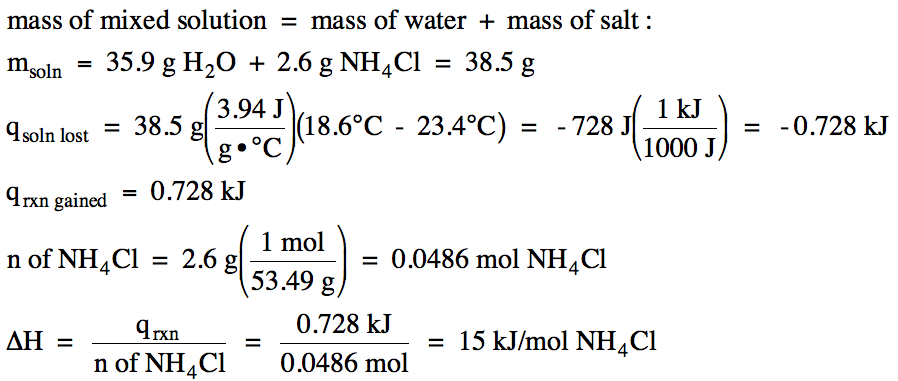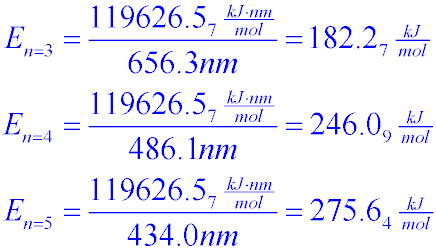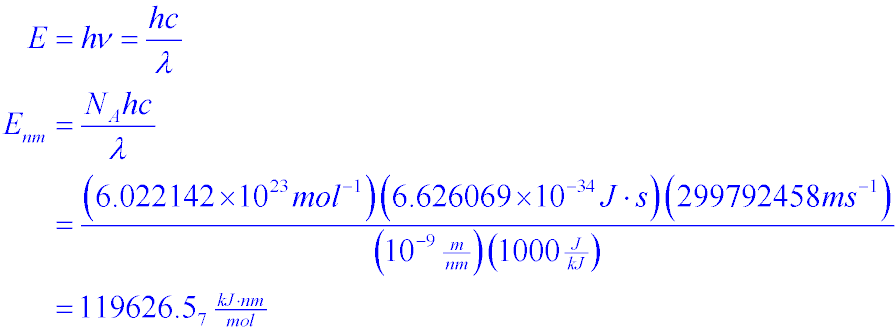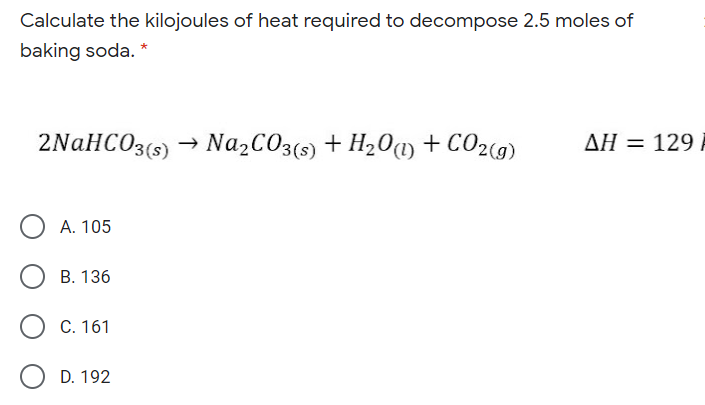How to calculate Energy yield in terms of KJ/L/d during bio-hydrogen production. (highlighted in red circle)? | ResearchGate

Daily Intake Guide: Healthy eating, made easy. Front-of-pack labelling for food and drink in Australia. - Calculating Food Energy

Calculate the energy in kilojoules per mole of electronic charge accelerated by a potantial of `... - YouTube
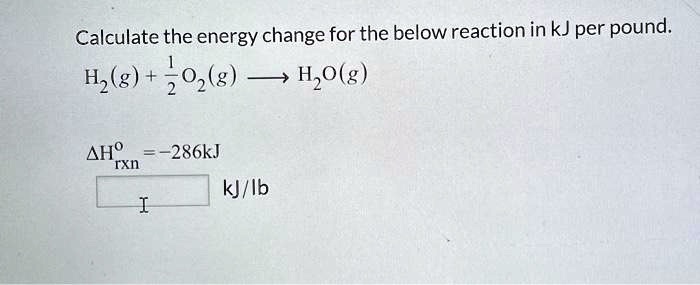
SOLVED: Calculate the energy change for the below reaction in kJ per pound: Hz (g) 02 (g) H,O(g) AHO TXn -286kJ kJ/lb
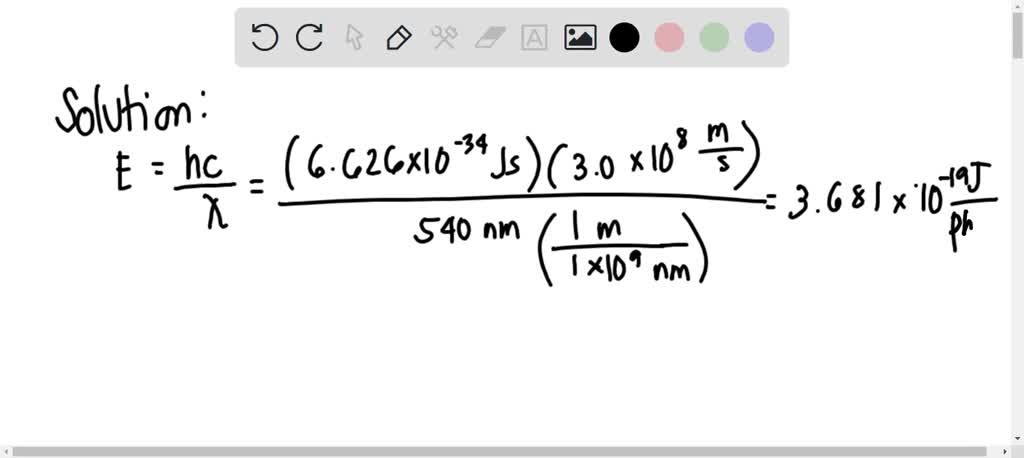
SOLVED: Calculate the energy in kJ/mole for a photon of light with wavelength 540. nm if n = 1. Group of answer choices 7.18x10-28 kJ/mole 6.46x10-11 kJ/mole 4.51 kJ/mole 222 kJ/mole 1.47